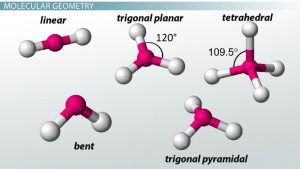
  Since this molecule has a steric number of 4 but is only bonded to 3 atoms, we can infer a trigonal pyramid geometry. The molecular geometry is T-shaped with bond angles of slightly less than 120° and slightly less than 90°. When there are two lone pairs (m3, n2 or AX 3 E 2), each lone pair occupies one of the three equatorial positions. The order of increasing H2O, NH bond 3, angle PH3 in H2O < PH3 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
